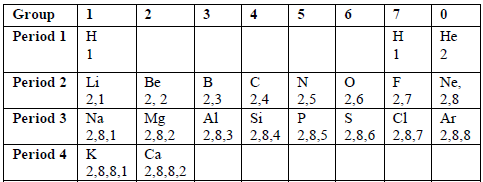
TOPIC 6: PERIODIC CLASSIFICATION | CHEMISTRY FORM 2
TOPIC 6: PERIODIC CLASSIFICATION | CHEMISTRY FORM 2
Constructing the modern periodic table has been a major scientific achievement. The first steps towards working out this table were taken long before anyone had any idea about the structure of atoms. The number of elements discovered increased steadily during the 19th century. Chemists began to find out patterns in their properties.
The Law of Triads
In 1817, the German scientist Johann Dobereiner noticed that calcium,
strontium and barium had similar properties, and that the atomic weight
of strontium was halfway between the other two. He found the same
pattern with chlorine, bromine and iodine and also with lithium, sodium
and potassium.
of strontium was halfway between the other two. He found the same
pattern with chlorine, bromine and iodine and also with lithium, sodium
and potassium.
So, he put forward the law of Triads: “If elements are
arranged in groups of three in order of increasing atomic weights,
having similar properties, then the atomic weight of the middle element
is the arithmetic mean of the atomic weights of the other two elements”,
E.g.
arranged in groups of three in order of increasing atomic weights,
having similar properties, then the atomic weight of the middle element
is the arithmetic mean of the atomic weights of the other two elements”,
E.g.
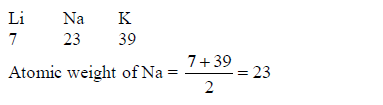
The
following are examples of Dobereiner’s triads:(Lithium, Sodium and
Potassium)(Calcium, Strontium and Barium)(Chlorine, Bromine and Iodine)
and(Iron, Cobalt and Nickel)
following are examples of Dobereiner’s triads:(Lithium, Sodium and
Potassium)(Calcium, Strontium and Barium)(Chlorine, Bromine and Iodine)
and(Iron, Cobalt and Nickel)
The Law of Octaves
In
1863 John Newlands, an English chemist noted that there were many pairs
of similar elements. In each pair, the atomic weights differed by a
multiple of 8. So, he produced a table with the elements in order of
increasing atomic weights, and put forward the Law of Octaves: “If
elements are arranged in order of their increasing atomic weights, the
properties of the 8th element, starting from a given one, are a kind of
repetition of the first element”.
1863 John Newlands, an English chemist noted that there were many pairs
of similar elements. In each pair, the atomic weights differed by a
multiple of 8. So, he produced a table with the elements in order of
increasing atomic weights, and put forward the Law of Octaves: “If
elements are arranged in order of their increasing atomic weights, the
properties of the 8th element, starting from a given one, are a kind of
repetition of the first element”.
This finding was comparable to the 8th note of music, hence the use of the word “octave”.
This
was the first table to show a periodic or repeating pattern of
properties. But it was not widely accepted because there were too many
inconsistencies. For example, he put copper and sodium in the same
group, even though have very different properties. Also iron was placed
in the same group as oxygen and sulphur.
was the first table to show a periodic or repeating pattern of
properties. But it was not widely accepted because there were too many
inconsistencies. For example, he put copper and sodium in the same
group, even though have very different properties. Also iron was placed
in the same group as oxygen and sulphur.
The Periodic Law
Dmitri
Mendeleev was born in Siberia, Russia, in 1834. By the time he was 32,
he was a professor of Chemistry. In 1869 Mendeleev advanced the work
done by Newlands and contributed very useful new ideas. He began by
listing all the known elements in order of increasing atomic mass. He
spotted that elements with similar properties appear at regular
intervals or periods down the list. His findings were the basis for the
Periodic Law: “The properties of elements are a periodic function of
their atomic masses”.
Mendeleev was born in Siberia, Russia, in 1834. By the time he was 32,
he was a professor of Chemistry. In 1869 Mendeleev advanced the work
done by Newlands and contributed very useful new ideas. He began by
listing all the known elements in order of increasing atomic mass. He
spotted that elements with similar properties appear at regular
intervals or periods down the list. His findings were the basis for the
Periodic Law: “The properties of elements are a periodic function of
their atomic masses”.
Mendeleev
placed similar elements into groups. He realized that not all elements
had been discovered. So he left gaps for new ones in the correct places
in his table. He also swapped the order of some elements to make them
fit better. He predicted the properties of the missing elements from the
properties of the elements above and below them in the table. He also
listed separately some elements which did not appear to fit into any
group i.e. iron, cobalt, nickel, etc.
placed similar elements into groups. He realized that not all elements
had been discovered. So he left gaps for new ones in the correct places
in his table. He also swapped the order of some elements to make them
fit better. He predicted the properties of the missing elements from the
properties of the elements above and below them in the table. He also
listed separately some elements which did not appear to fit into any
group i.e. iron, cobalt, nickel, etc.
Table 6.1: Mendeleev’s short form of the Periodic Table
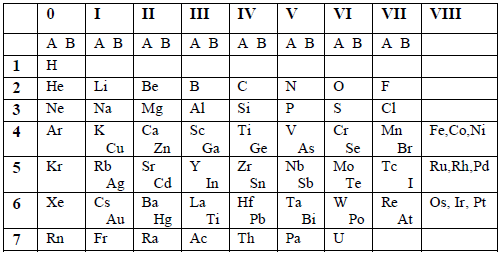
The
table had 9 vertical columns which he called Groups. The groups were
numbered from 0 to 8. The elements in group 0 were not known by then,
but were discovered later on. Groups 1 to 7 were subdivided into A and B
subgroups. Group 0 included the transition elements. Noble gases were
later placed in group 0.
table had 9 vertical columns which he called Groups. The groups were
numbered from 0 to 8. The elements in group 0 were not known by then,
but were discovered later on. Groups 1 to 7 were subdivided into A and B
subgroups. Group 0 included the transition elements. Noble gases were
later placed in group 0.
There
were 7 horizontal rows which he called periods. All vacant positions in
the table stood for new elements yet to be discovered.
were 7 horizontal rows which he called periods. All vacant positions in
the table stood for new elements yet to be discovered.
Usefulness of Mendeleev’s classification
- The table summarized a large amount of information about the elements based on their chemical properties.
- The
table was very useful in predicting the existence and properties of
undiscovered elements, for which gaps had been left in the table. - The table was also used in checking relative atomic masses of elements.
Limitations of Mendeleev’s classification
- In
three cases, pairs of elements had to be included in one group based on
inverse order of their atomic weights so as to fit into groups of
elements having similar properties. These pairs were argon (39.9) and
potassium (39.1), cobalt (58.9) and nickel (58.9); plus tellurium
(127.5) and iodine (126.9). This difficulty was resolved when the basis
of classification was based on the atomic number instead of the atomic
mass. - The elements that were placed in group VIII formed an incompatible mixture.
- The placing of two different families in one group e.g. K and Cu; Ca and Zn, etc.
The
periodic table is the chemists map. It helps you understand the
patterns in chemistry. Today we take it for granted. But it took
hundreds of years, and work of hundreds of chemists, to develop.
periodic table is the chemists map. It helps you understand the
patterns in chemistry. Today we take it for granted. But it took
hundreds of years, and work of hundreds of chemists, to develop.
The
Modern Periodic Table is similar to that of Mendeleev, but contains
several improvements. Elements are arranged in order of atomic number
instead of atomic mass. This means that elements no longer have to swap
places to fit correctly. Many new elements have been discovered and
slotted into the spaces left by Mendeleev. Also metals and non-metals
are clearly separated. The Modern Periodic Table is shown in
Modern Periodic Table is similar to that of Mendeleev, but contains
several improvements. Elements are arranged in order of atomic number
instead of atomic mass. This means that elements no longer have to swap
places to fit correctly. Many new elements have been discovered and
slotted into the spaces left by Mendeleev. Also metals and non-metals
are clearly separated. The Modern Periodic Table is shown in
Figure 6.1.
Figure 6.1: The Modern Periodic Table
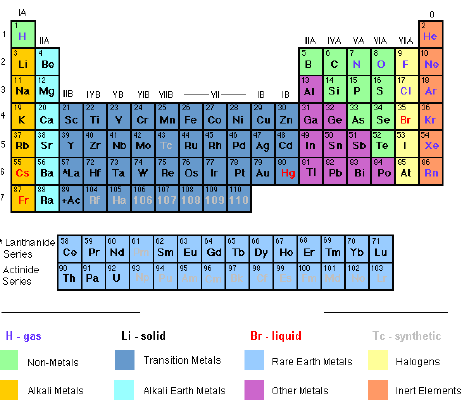
The
long form of the periodic table is the commonly used form of the
periodic table. The elements in the table are arranged based on their
atomic weights, starting from hydrogen (1), helium (2), lithium (3),
beryllium (4) and so on. The elements appear in vertical columns and
horizontal rows.
long form of the periodic table is the commonly used form of the
periodic table. The elements in the table are arranged based on their
atomic weights, starting from hydrogen (1), helium (2), lithium (3),
beryllium (4) and so on. The elements appear in vertical columns and
horizontal rows.
The
vertical columns in the table are called Groups, numbered I, II, III,
IV, V, VI, VII and 0, which is also known as group VIII. Group I
contains the elements lithium (L), sodium (Na), rubidium (Rb), caesium
(Cs) and francium (Fr). Group II consists of elements starting from
sodium (Na) down to radium (Ra). Some of the groups have special names.
vertical columns in the table are called Groups, numbered I, II, III,
IV, V, VI, VII and 0, which is also known as group VIII. Group I
contains the elements lithium (L), sodium (Na), rubidium (Rb), caesium
(Cs) and francium (Fr). Group II consists of elements starting from
sodium (Na) down to radium (Ra). Some of the groups have special names.
Group I is often called the alkali metals.
Group II the alkaline earth metals.
Group VII the halogens.
Group 0 the noble gases.
The
transition metals (or elements) form a separate block in the middle of
the periodic table between group II and III. The atoms of these elements
have more complicated electron arrangements. Note that the group
contains many common metals such as iron (Fe), Nickel (Ni), copper (Cu),
and Zinc (Zn). One of the interesting properties of these elements is
that they form coloured compounds.
transition metals (or elements) form a separate block in the middle of
the periodic table between group II and III. The atoms of these elements
have more complicated electron arrangements. Note that the group
contains many common metals such as iron (Fe), Nickel (Ni), copper (Cu),
and Zinc (Zn). One of the interesting properties of these elements is
that they form coloured compounds.
Main features of the Modern Periodic Table
- The elements in the table are placed in order of their atomic numbers instead of their atomic masses.
- There are a total of 18 groups and 7 periods.
- There are 5 blocks of similar elements in the periodic table as shown in figure 6.2.
- The
normal (non-transition) elements (groups 1-7) have their outermost
shells incomplete, meaning that they can allow additional electrons to
enter into their outermost orbital (valency shell). But each of their
inner shells is complete. - The transition metals have their outermost as well as their penultimate (second last) shells incomplete.
- Elements
of group 0 (noble gases) have their shells complete. These elements
show little reactivity. That is why they wereonce called „inert‟ gases
because they are very unreactive; or „rare gases‟ because they were
rarely found. - Gaps left by Mendeleev for undiscovered elements
(now occupied by the transition elements and the noble gases) have been
filled by the respective elements following their discovery. Man-made
elements have also found a place in the periodic table. - Metals
have been clearly separated from non-metals. Metalloids or semi metals
(poor metals) have also been included. Metalloids are elements whose
properties are intermediate between metals and non-metals. They include
boron (B), silicon (Si), germanium (Ge), arsenic (As), antimony (Sb) and
tellurium (Te). In some publications, germanium and antimony are
usually classed as poor metals and the rest as non-metals.